Silicone Biopolymer Removal
Choosing the Best Plastic Surgeon
Our office has two decades of experience in the removal of silicone, PMMA, and Biopolymer removal from the face and body. Dr. Raffy Karamanoukian has researched, lectured, and taught hundreds of plastic surgeons regarding his approach to less-invasive silicone removal from the face and buttocks. He is a Fellow of the American College of Surgeons, a diplomate of the American Board of Plastic and Reconstructive Surgeons, and a dedicated surgeon at Kare Plastic Surgery in Los Angeles.

Complications from liquid silicone injections have reached epidemic proportions. Our office sees patients from around the world who have undergone silicone injection in various parts of the body. The lips, hips, thighs, and buttocks are common areas of injection. Patients with silicone in their bodies may desire removal of the silicone because of unwanted swelling, redness, infection, inflammation, pain, discomfort, and ulceration. These are very difficult problems to solve and a regimented protocol is necessary to achieve acceptable results and maintain aesthetic contour.
There are many different types of fillers that are used for permanent volumization of the soft tissue. The most common types include silicone biopolymer, PMMA Bellafill, and Artefill. Patients seek treatment at Kare Plastic Surgery in Santa Monica and Beverly Hills when symptoms arise from silicone injections. The most common signs and symptoms that are reported to our office include the following:
- Erythema (Redness) and Cellulitis are signs that there may be a subclinical or true infection within the soft tissue. Patients may experience warmth in the area, redness, and signs fo infection.
- Generalized firmness of the soft tissue, or hard lumps underneath the skin.
- Unrelenting pain, itching, and burning that does not subside with position.
- Numbness in the area of injection, or down the legs.
- Red or brown skin discoloration.

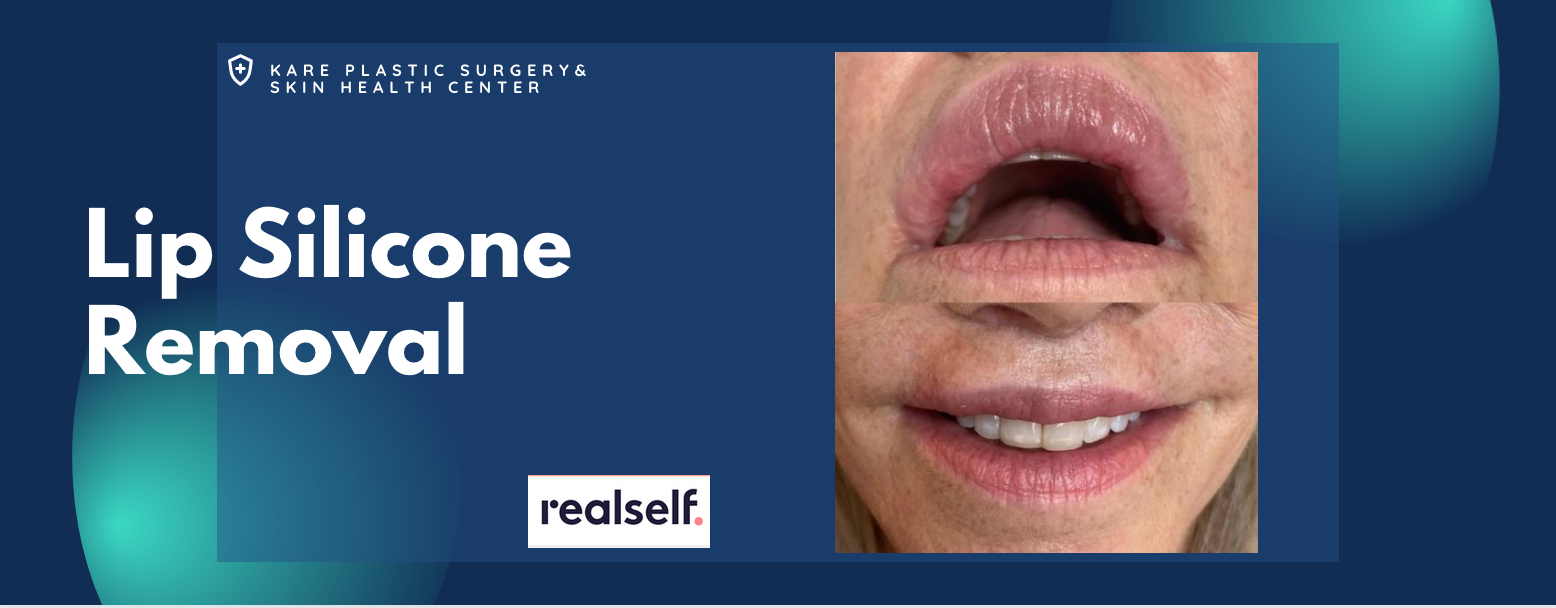
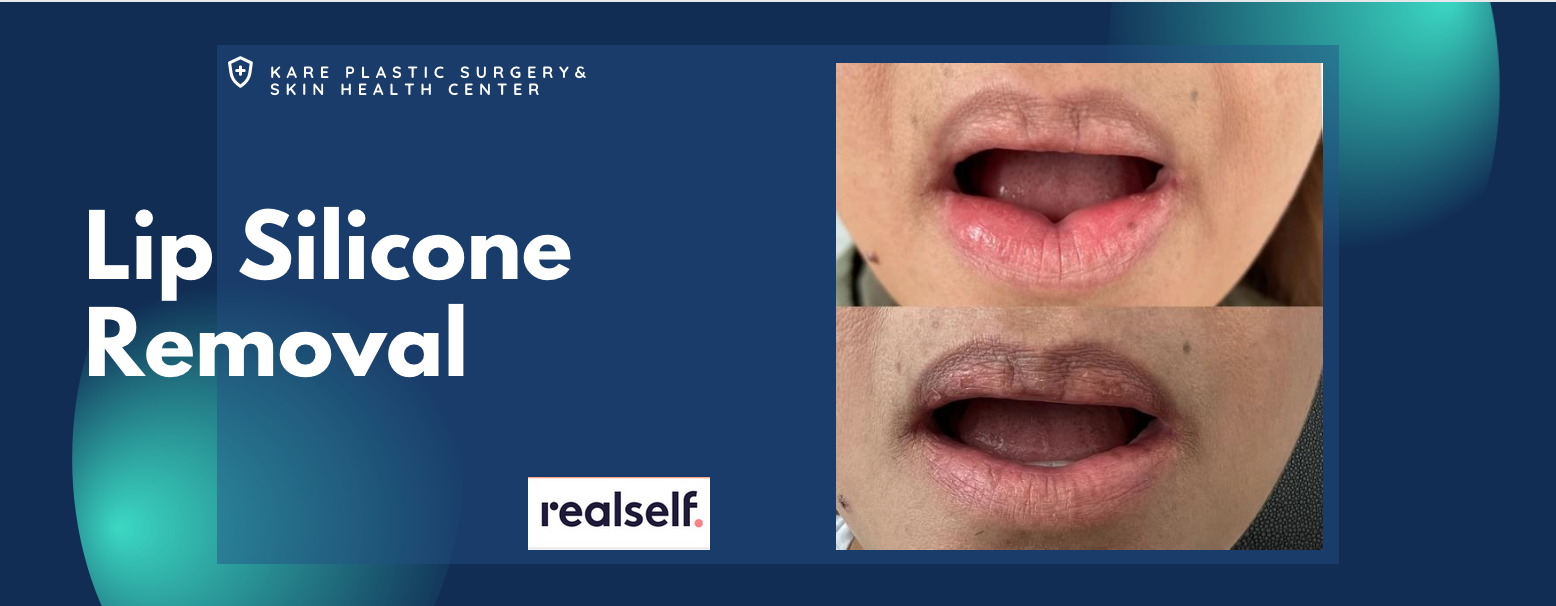
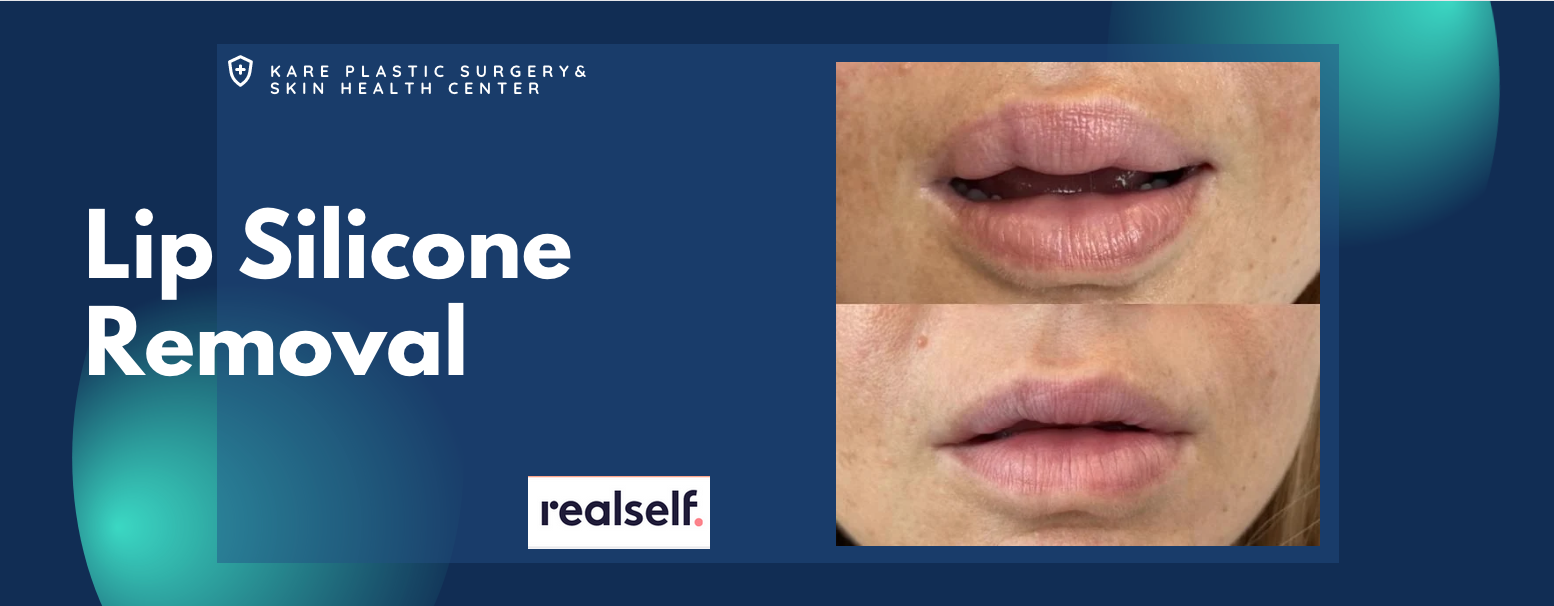
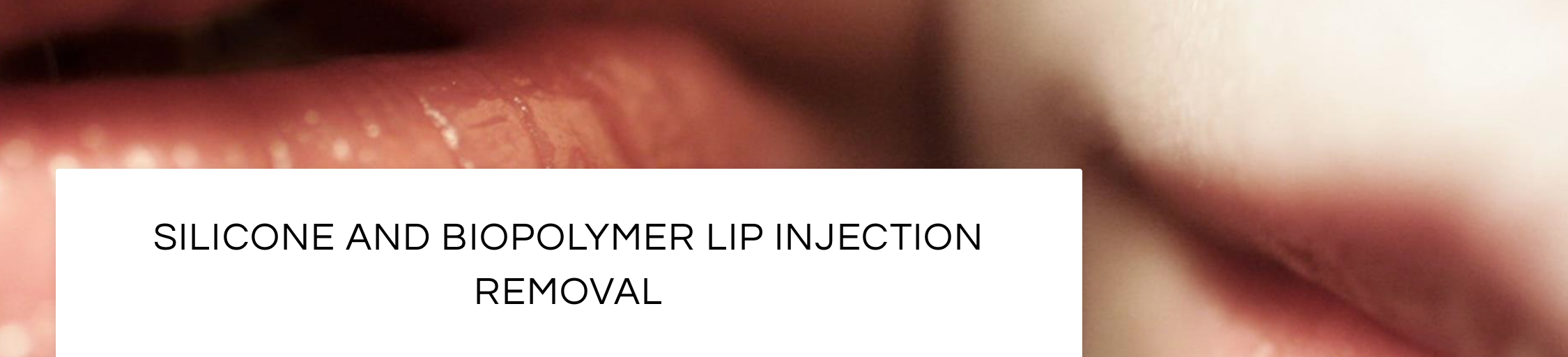
Removal of Silicone from the Lips
Silicone is often injected into the upper and lower lip areas. Unlike temporary lip augmentation with Juvederm or Restylane, liquid silicone does not dissolve with time. On the contrary, silicone is a permanent filler that not only stays in the body, but often develops into bigger inflammatory nodules that are known as granulomas. Some patients will also develop non-nodular swelling, itching, burning, and pain that is directly attributed to the silicone itself.
The treatment of lip silicone or lip nodules is difficult. Treatment begins with the injection of corticosteroids to decrease the inflammatory effects of the silicone itself. These injections should be performed by a physician who is familiar with the treatment of silicone in the lips, as complications can occur. In practice, silicone that is minimally-responsive to corticosteroids can be surgically removed using a variety of techniques. These procedures are usually performed in an outpatient surgical suite under anesthesia.
Dr. Raffy Karamanoukian is an expert in the treatment of silicone injection complications. He performs surgery for silicone nodules, lumps, and silicone complications using surgical and non-surgical methods. Patients who have non-surgical silicone nodules may benefit from our intralesional injections that can help reduce the volume and visibility of filler.

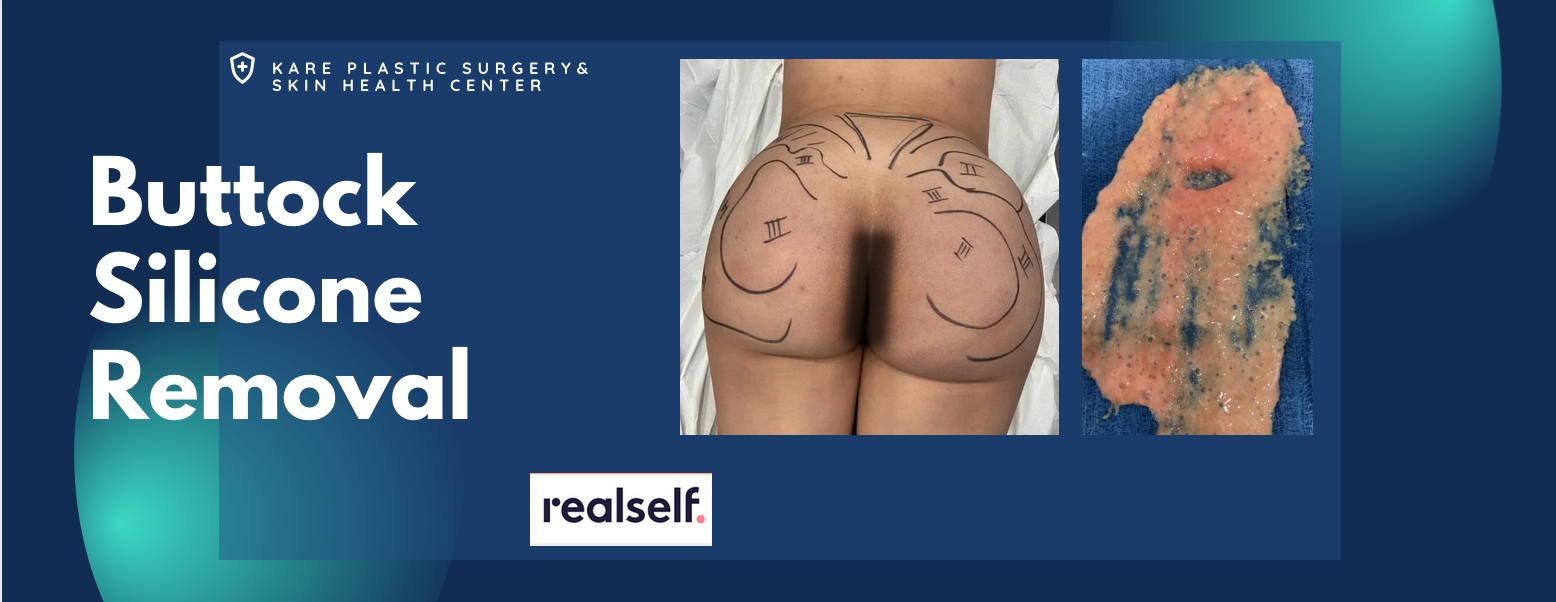
Removal of Silicone Fillers from the Buttocks
Silicone injections are commonly injected into the soft tissue of the buttocks and hips in order to enhance the contour and volume of the lower torso, buttocks, and thighs. Women who desire a Brazilian Butt Lift may opt for silicone injections if there is no available fat to harvest as donor tissue for fat transfer; but fail to appreciate the longterm risk of soft tissue inflammation and granuloma formation.

The removal of liquid silicone from the buttocks is possible if there is limited fibrosis of the soft tissue. Prolonged inflammation may damage the underlying vascularity of the soft tissue, making it increasingly difficult to safely remove the liquid silicone after extended periods of time. If you have liquid silicone injections in the buttocks or hips, it is imperative to seek early treatment to mitigate the longterm effects of chronic inflammation, soft tissue fibrosis, granuloma formation, and scarring.
Dr. Raffy Karamanoukian is renowned for his surgical protocols to remove silicone from soft tissue. He has developed the Karamanoukian Soft Tissue Grading Scale for soft tissue reaction following the administration of silicone injections. If you are experiencing problems associated with liquid silicone injections into the buttocks, a consultation can help you understand your options for treatment.

Silicone and other biopolymers used for soft tissue augmentation can cause a chronic inflammatory reaction that induces an inflammatory autoimmune response. Treating the biopolymers without effective removal is not a long-term treatment plan as the autoimmune inflammatory reaction will likely recur and cause persistent changes in the skin and soft tissue. These changes include scar formation, tissue fibrosis, edema, and swelling, hypervascularity, skin color, changes, and sagging skin. For these reasons, Dr. Karamanoukian has developed a lesson basic modality for removal of unwanted silicone biopolymers without extensive surgery.
Removal of Silicone from the Face
Although silicone oils (Biogel, Biopolimero, Bioplastique, and Biosil) can be injected anywhere on the face and body, it becomes more and more complicated to remove the silicone from aesthetically-important areas such as the nose, eyes, cheeks, and chin. Surgical removal of silicone is further complicated by the anatomic restrictions imposed by important neurovascular structures that can be injured if the silicone is removed.
Dr. Karamanoukian is a leading expert in the removal of silicone from the face and body. During your consultation, he will evaluate the Karamanoukian Grading Scale for soft tissue reaction to determine if there are surgically-feasible options for silicone removal. In some cases, the risk of silicone removal may outweigh the benefits; making it exceedingly difficult to justify the removal of silicone in an elective setting. In cases of symptomatic granuloma formation, ulceration, chronic tissue fibrosis, and infection, there may be a medical justification for emergent removal.
Silicone Filler Migration
Silicone Biopolymers can migrate from one zone of the body to another. Understanding permanent filler migration patterns is not simple, because there are many different types of permanent fillers and many different viscosities of filler that must be analyzed. In most cases, we see silicone hydrogel and silicone biopolymer migration across tissue planes on the buttocks and body. These fillers can move across tissue planes, but generally are found within the subcutaneous fat and peri-fascial planes.
Dr. Karamanoukian is an expert in the removal of permanent filler from the body and can help you understand and predict patterns of soft tissue filler migration. . Learn more
Removal of Artecoll
We are often asked to remove Artecoll products from the face, especially around the eyes and the marionette lines of the lower face. Artecoll filler can incite a dense inflammatory reaction leading to granulomas, or hard nodules within the skin. It is often difficult to differentiate Artecoll granulomas from normal tissue because there is a less defined interface between the two. In some cases, hard nodules can be removed by Dr. Raffy Karamanoukian using defined protocols for granuloma clearance. Surgical options remain the gold standard for treatment.
Cases of symptomatic granulomas from Artecoll or Artefill can be managed with non-surgical options, including intralesional therapy. A consultation at Kare Plastic Surgery + Skin Health Center can help you define your options.
Removal of Bellafill
Bellafill is an FDA-approved filler formulated to treat nasolabial folds and moderate to severe atrophic facial acne scars. Our Santa Monica plastic surgery office specializes in the removal of Bellafill and Bellafill granulomas from the face using a surgical approach. Bellafill is composed of collagen and polymethylmethacrylate (PMMA) and can cause granulomas and scarring. Dr. Raffy Karamanoukian can help formulate a surgical plan that removes the filler with minimal complication and risk. Unwanted Bellafill may cause nodules and persistent edema.
There are several types of fillers that can react similarly to Bellafill and these include Artefill, Artecoll, PMMA. The filler polymethyl methacrylate contains microspheres that can contain varied amounts of collagen and are used to fill volume into soft tissue and skin. According to the manufacturers of Bellafill, the "collagen gel in Bellafill® provides immediate volume and lift to correct wrinkles and acne scars. Once injected, these microspheres stay in place and create a base that provides structural support for smoother-looking skin." Although this is true, it may also cause reactions in a small subset of patients.
Bellafill contains polymethylmethacrylate (PMMA) microspheres that are used for soft-tissue augmentation. Bellafill injections are usually placed superficially in the lips, cheeks, chin, and jawline and can sometimes cause severe, long-term complications including filler migration, granuloma formation, nodules, swelling, edema, and uneven contour. These are common side-effects of Bellafill. If you have had Bellafill PMMA microspheres injected into your lips or face, you may require plastic surgery treatment by a board-certified specialist.
Dr. Raffy Karamanoukian is a world leader in the management of Bellafill and PMMA complications in the face and body. There are several different methods of treatment for Bellafill complications, and our Los Angeles plastic surgery center is equipped with the latest technology and treatment options to help you regain your confidence after poorly placed PMMA injections. Dr. Karamanoukian is a fellow of the American College of Surgeons and an active member of the American Society of Plastic Surgeons. He is a diplomat of the American Board of Plastic and Reconstructive Surgery and can help you manage complications from Bellafill or other PMMA injections.
If you are looking for a treatment option for PMMA in the lips or face, there are two main treatment options available in our center. Although surgical removal of Bellafill and PMMA is a common method of repair, it often requires placement of incisions directly above the nodules in the face. Surgical excision of PMMA is a good option when the incisions can be hidden in barely noticeable areas of the face. Diffuse concentrations of PMMA Bellafill are unlikely to resolve with surgical excision, and thus the second option involves intralesional injections to reduce the fibrosis and scar tissue around the PMMA ,
Intralesional injections are a newer option for people who suffer from complications associated with Bellafill. This method is non-surgical and less invasive and is appropriate for patients who have diffuse concentrations of Bellafill in areas that are cosmetically-sensitive and less likely to improve with surgical excision. Intralesional injections allow our board-certified plastic surgeon to reduce the size and density of PMMA into smaller pieces that can be managed with less complications.
Surgical removal and intralesional injection removal of Bellafill can cause some side effects, such as mild, bruising and swelling; but these are usually transient effects that can be improved with time using laser technology.
Your options for removing Bellafill from the lips and face will depend on the results of your consultation and examination. Dr. Karamanoukian will perform biometric measurements of tissue volume and PMMA volume during your consultation in Los Angeles. Depending on the severity of your issues, we can offer options that can reduce scarring to the best ability.
Surgical removal of Bellafill is the most common option and involves an excisional technique in which a small incision is placed above nodules under local anesthesia or general anesthesia. If your PMMA is diffuse and non-focalized, incision may not be the best option for you. Surgical excision followed by suture repair can provide you with an effective solution to remove PMMA permanently. Patients can tolerate PMMA surgical excision very well and often this is performed in our outpatient Los Angeles plastic surgery center.
Intralesional injections of PMMA utilise medicines to break up the fibrosis and scar tissue around PMMA nodules. There are no definitive measures to dissolve the actual PMMA microspheres, but a major treatment option is to break the fibrosis and nodular granuloma formation in order to make it less cosmetically visible. This is an excellent option for most patients and can give you cosmetic and medical relief from poorly placed Bellafill injections.
Our Los Angeles plastic surgery office is also a major treatment center for Bellafill injections that are placed for acne scars. Bellafill injections in the epidermis and dermis can form nodular lesions that cause permanent deformity. Dr. Karamanoukian is an expert in Bellafill removal for the skin.
Removal of Bioplastique
Our office has had several patients who have undergone successful removal of Bioplastique from the soft tissue of the lips and face. This particular filler is often of questionable provenance and thus has variable complication profiles. Bioplastique silicone can be removed from the lips using Dr. Raffy Karamanoukian's pioneering techniques. Surgical removal of Bioplastique from the soft tissue of the face is limited by anatomy, particularly neurovascular structures that may be at risk during surgical excision.
Removal of Biopolimero
Biopolimero, Biopolimer, Biogel, and Biopolymers are a group of non-FDA approved fillers that are used for soft tissue volume augmentation on the lips, face, and body. Our office consults on Biopolymer injections to the buttocks and face; and our expert plastic surgeon, Dr. Raffy Karamanoukian, has developed a protocol to remove unwanted Biopolymers from the soft tissue.
Removal of Dermagen
Dermagen is a mineral oil/silicone polymer that has been used for soft tissue augmentation of the face and body. Removal of Dermagen should be initiated early, as longterm exposure to Dermagen within the soft tissue can lead to chronic inflammation, granulomatous reaction, and soft tissue fibrosis. Dr. Raffy Karamanoukian is an expert in Dermagen removal, if possible, from the face and body. Surgical removal of Dermagen is limited to those areas where critical nerves are not compromised during treatment. Although Dermagen and Biopolymers have been used in soft tissue augmentation, longterm symptoms of pain, discomfort, infection, foreign body reaction, and inflammation have been reported.
Removal of Juvederm or Restylane
Juvederm and Restylane Hyaluronic acid facial fillers can be readily removed using Hyaluronidase enzymes Vitrase and Hylenex which dissolve the injected material using enzymatic activity. Unlike silicone and Artefill injections hyaluronic acid is reversible without surgery. A small subset of our patients, however, do not respond to treatment because the material is a more viscous form of hyaluronic acid or not Juvederm or Restylane at all.
